2026
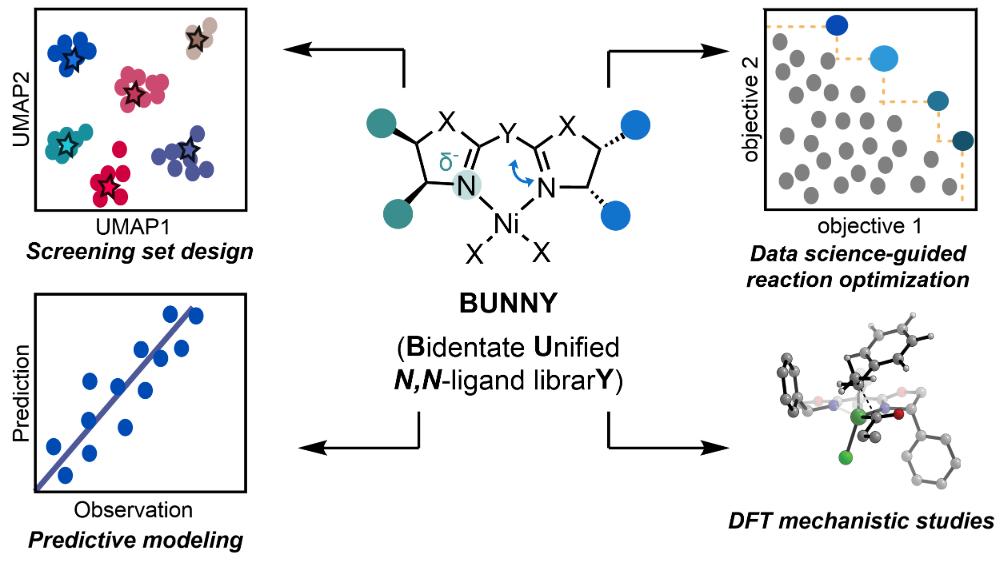
111. “BUNNY: An N,N-Bidentate Nitrogen Ligand Descriptor Library–Development and Application to Modeling of Ni-Catalyzed Asymmetric Cross-Electrophile Coupling Reactions” Gutiérrez Valencia, N. E.; Schleinitz, J.; Wild, T. H.; Williams, W. L.; Mantin, D.; Doyle, A. G.; Reisman, S. E.; Sigman, M. S. ACS Catal., 2026, ASAP. [DOI: 10.1021/acscatal.6c02585] Link PDF
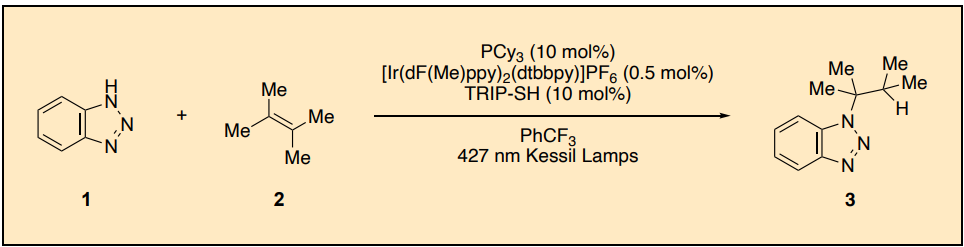
110. “Intermolecular Hydroamination of Benzotriazole with Tetramethylethylene” Fan, F.; Wearing, E. R.; Loughney, C. B.; Doyle, A. G. Org. Synth., 2026, 103, 121-134. [DOI: 10.15227/orgsyn.103.0121] Link PDF
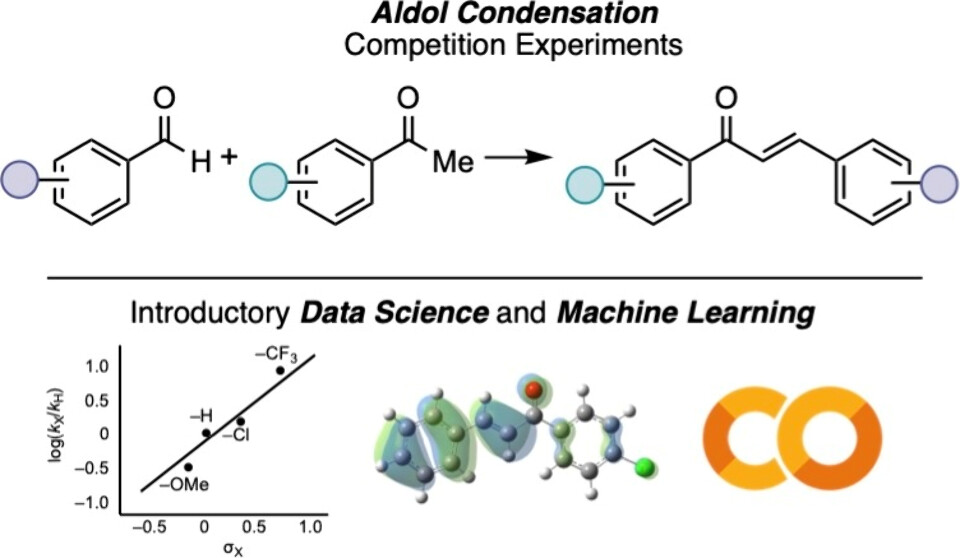
109. “Integrating Data Science and Machine Learning with an Aldol Condensation Laboratory” Min, D. S.; Fan, F.; Doyle, A. G. J. Chem. Educ., 2026, 103, 1497-1504. [DOI: 10.1021/acs.jchemed.5c00994] Link PDF
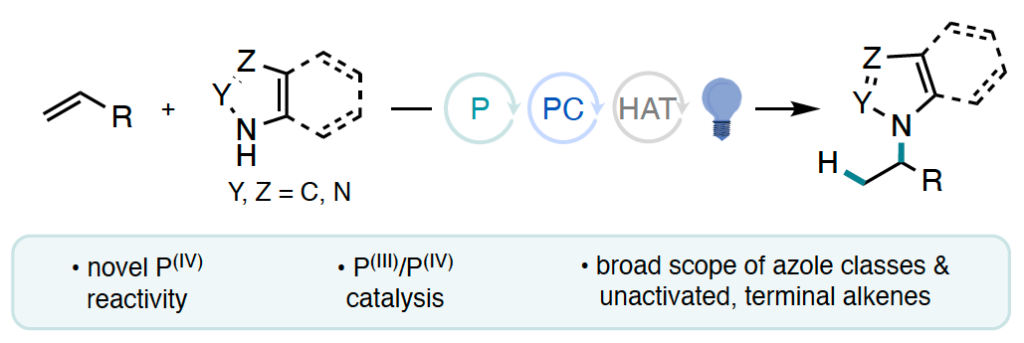
108. “Markovnikov hydroamination of terminal alkenes via phosphine redox catalysis” Fan, F.; Sedillo, K. F.; Maertens, A. J.; Doyle, A. G. Nature, 2026, 652, 96–104. [DOI: 10.1038/s41586-026-10263-7] Link PDF
107. “Transferable enantioselectivity models from sparse data” Gallarati, S.; Bucci, E. M.; Doyle, A. G.; Sigman, M. S. Nature, 2026, 651, 637-646. [DOI: 10.1038/s41586-026-10239-7] Link PDF

106. “Synergizing Chemical and AI Communities for Advancing Laboratories of the Future” Oh, S.; Fang, X.; Lin, I.-H.; Dee, P.; Dunham, C. S.; Copp, S. M.; Doyle, A. G.; de Alaniz, J. R.; Gu, M. ACS Cent. Sci., 2026, 12, 157-173. [DOI: 10.1021/acscentsci.5c01994] Link PDF 100th paper!
2025
105. “Development, Application, and Mechanistic Interrogation of a Dual Ni Catalysis Approach to Photoredox-Based C(sp3)–C(sp3) Cross-Coupling” Bucci, E. M.; Perea, M. A.; Lalisse, R. F.; Mukherjee, P.; Raab, T. J.; Kannadi Valloli, L.; Min, D. S.; Bird, M. J.; Gutierrez, O.; Doyle, A. G. J. Am. Chem. Soc., 2025, 147, 42311–42329. [DOI: 10.1021/jacs.5c11247] Link PDF

104. “Multi-level QTAIM-Enriched Graph Neural Networks for Resolving Properties of Transition Metal Complexes” Gee, W.; Doyle, A. G.; Vargas, S.; Alexandrova, A. Digital Discovery, 2025, 4, 3378–3388. [DOI:10.1039/D5DD00220F] Link PDF
103. “Considerations in Pursuing Reaction Scope Generality” Zacate, S. B.; Dantas, J. A.; Lin, S.; Doyle, A. G.; Sigman, M. S. Angew. Chem. Int. Ed., 2025, 64, e202511091. [DOI:10.1002/anie.202511091] Link PDF
102. “Reactivity Studies of Bipyridine-Ligated Nickel(I) and Nickel(0) Complexes Inform the Mechanism in Modern Cross-Coupling Reactions” Raab, T. Judah.; Doyle, A. G. J. Am. Chem. Soc., 2025, 147, 33991–34000. [DOI: 10.1021/jacs.5c11247] Link PDF
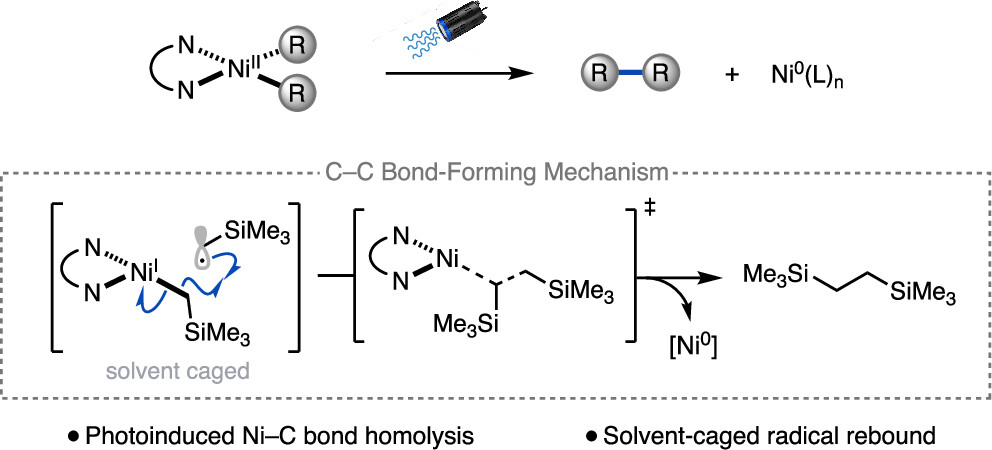
101. “Light-Promoted C(sp³)–C(sp³) Reductive Elimination from Dialkyl Ni(II) Complexes” Cusumano, A. Q.; Chaffin, B. C.; Cagan, D. A.; DiLuzio, S.; Sutcliffe, E.; Hadt, R. G.; Doyle, A. G. J. Am. Chem. Soc., 2025, 147, 32941–32950. [DOI: 10.1021/jacs.5c09925] Link PDF
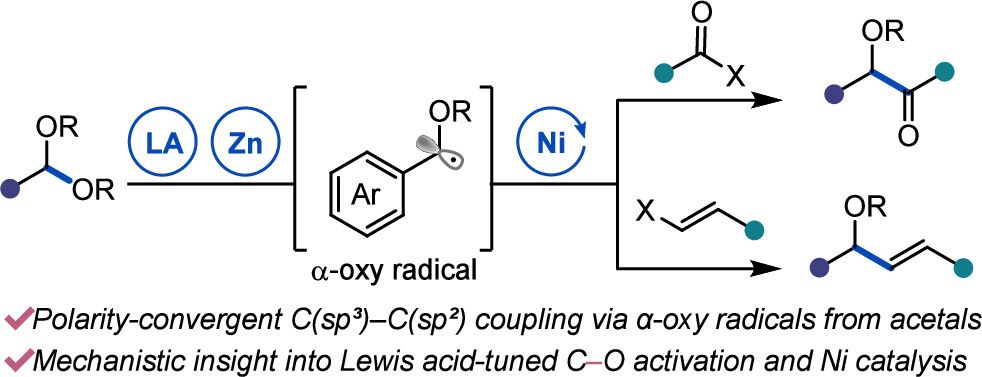
100. “Ni-Catalyzed Reductive Coupling of Acetals with Anhydrides and Vinyl Triflates via Single-Electron C–O Activation” Kim, E.; Borden, M. A.; Hwang, J.; Doyle, A. G.; Dongbang, S. Org. Lett., 2025, 27, 9454–9459. [DOI: 10.1021/acs.orglett.5c02788] Link PDF
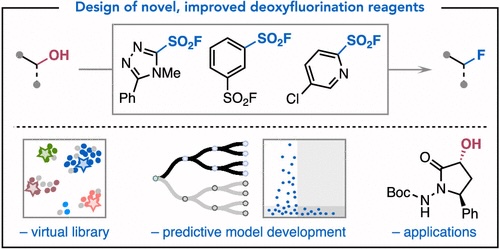
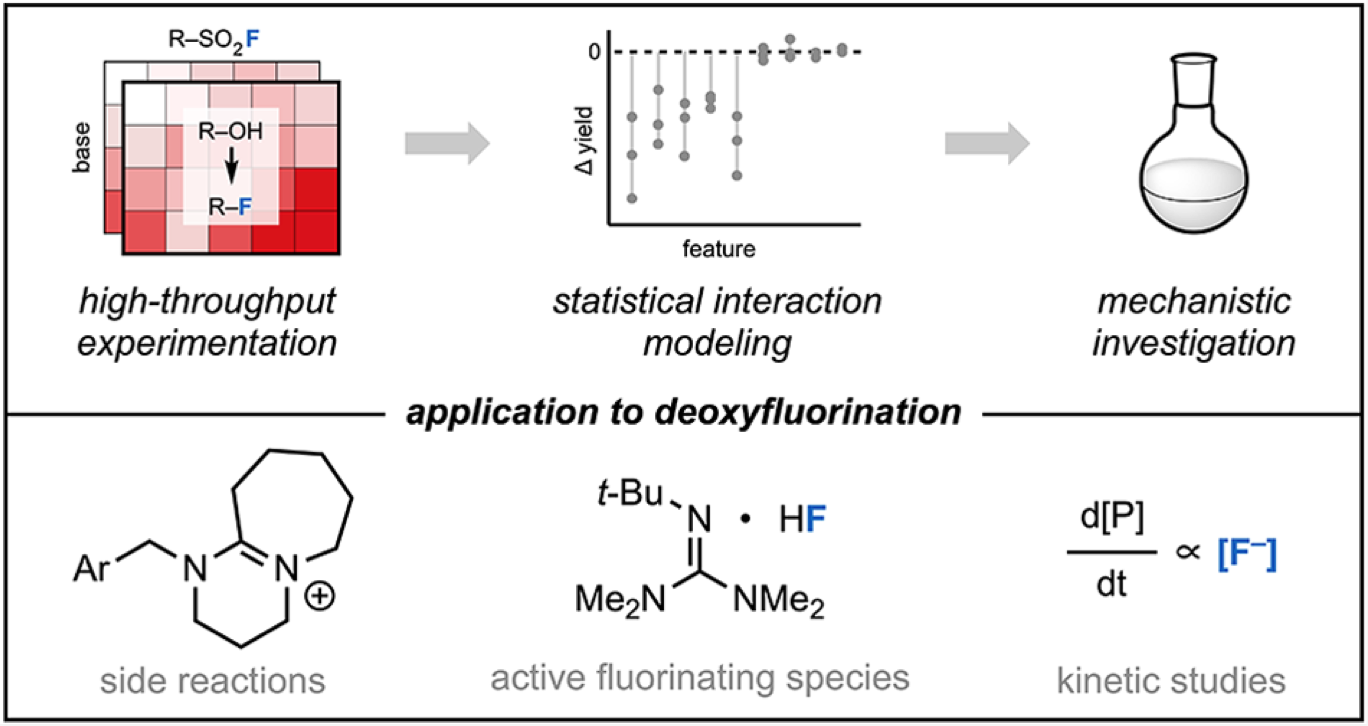
99. “Data Science-Guided Development of Deoxyfluorination Reagents with Enhanced Reactivity, Practicality, and Safety” Ruos, M. R.; Romer, N. P.; Deichert, J. A.; Alabanza, L. M.; Gandhi, S. S.; Brown, G. Z.; Walroth, R. C.; Cruz, K.; Gosselin, F.; Hong, A. Y.; Sigman, M. S.; Doyle, A. G. J. Am. Chem. Soc., 2025, 147, 25815–25824. [DOI: 10.1021/jacs.5c07548] Link PDF
98. “Fabrication of SERS Substrates Using Silver-Coated Gold Nanostars for Chemical Sensing: A Multiobjective Bayesian Optimization Approach” Giordano, A. N.; Franqui-Rios, S.; Quarin, S. M.; Vang, D.; Austin, D. R.; Doyle, A. G.; Baldwin, L. A.; Strobbia, P.; Rao, R. ACS Appl. Nano Mater., 2025, 8, 11930-11939. [DOI: 10.1021/acsanm.5c01462] Link PDF

97. “Applying Active Learning toward Building a Generalizable Model for Ni-Photoredox Cross-Electrophile Coupling of Aryl and Alkyl Bromides” Souza, L. W.; Ricke, N. D.; Chaffin, B. C.; Fortunato, M. E.; Jiang, S.; Soylu, C.; Caya, T. C.; Lau, S. H.; Wieser, K. A.; Doyle, A. G.; Tan, K. L. J. Am. Chem. Soc., 2025, 147, 18747-18759. [DOI: 10.1021/jacs.5c02218] Link PDF

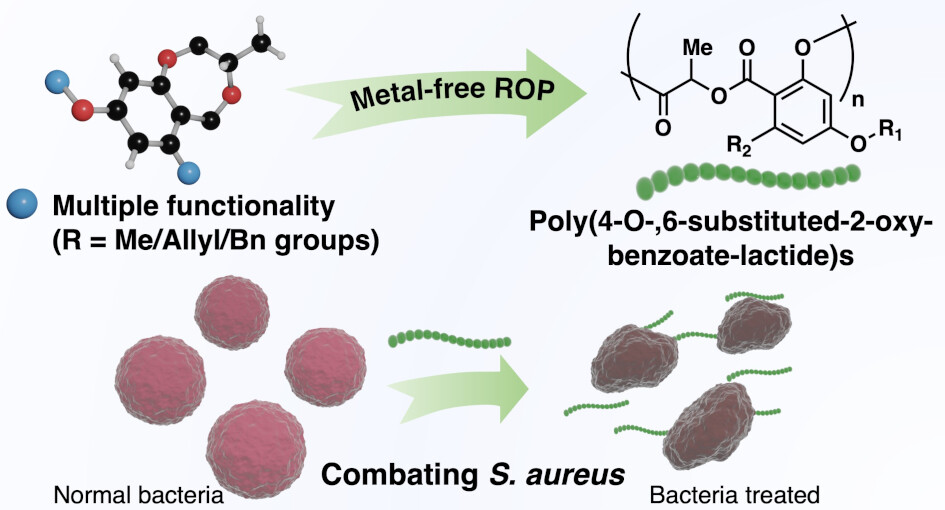
96. “Biosourced Functional Hydroxybenzoate-co-Lactide Polymers with Antimicrobial Activity” Salas-Ambrosio, P.; Vexler, S.; Sivasankaran, R. P.; Vlahakis, N.; Lai, R. S.; Johnson, C.; Baas-Maynard, S. I.; Min, D. S.; Lower, H.; Doyle, A. G.; Tang, Y.; Rodriguez, J. A.; Chen, I. A.; Read de Alaniz, J.; Maynard, H. D. J. Am. Chem. Soc., 2025, 147, 19230-19238. [DOI: 10.1021/jacs.5c04624] Link PDF
95. “Diversification of acridinium photocatalysts: Property tuning and reactivity in model reactions” Wang, J. Y.; Fan, F.; Ruos, M. E.; Adao Gomes, L.; Lavin, M.; O’Connor, T. J.; Lopez, S. A.; Doyle, A. G. Tet. Lett., 2025, 160, 155546 [DOI: 10.1016/j.tetlet.2025.155546] Link PDF
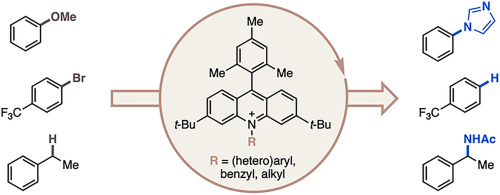
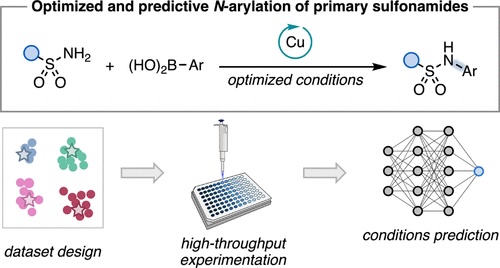
94. “Data Science-Driven Discovery of Optimal Conditions and a Condition-Selection Model for the Chan–Lam Coupling of Primary Sulfonamides” Gandhi, S. S.; Brown, G. Z.; Aikonen, S.; Compton, J. S.; Neves, P.; Martinez Alvarado, J. I.; Strambeanu, I. I.; Leonard, K. A.; Doyle, A. G. ACS Catal., 2025, 15, 2292-2304. [DOI: 10.1021/acscatal.4c07972] Link PDF
2024
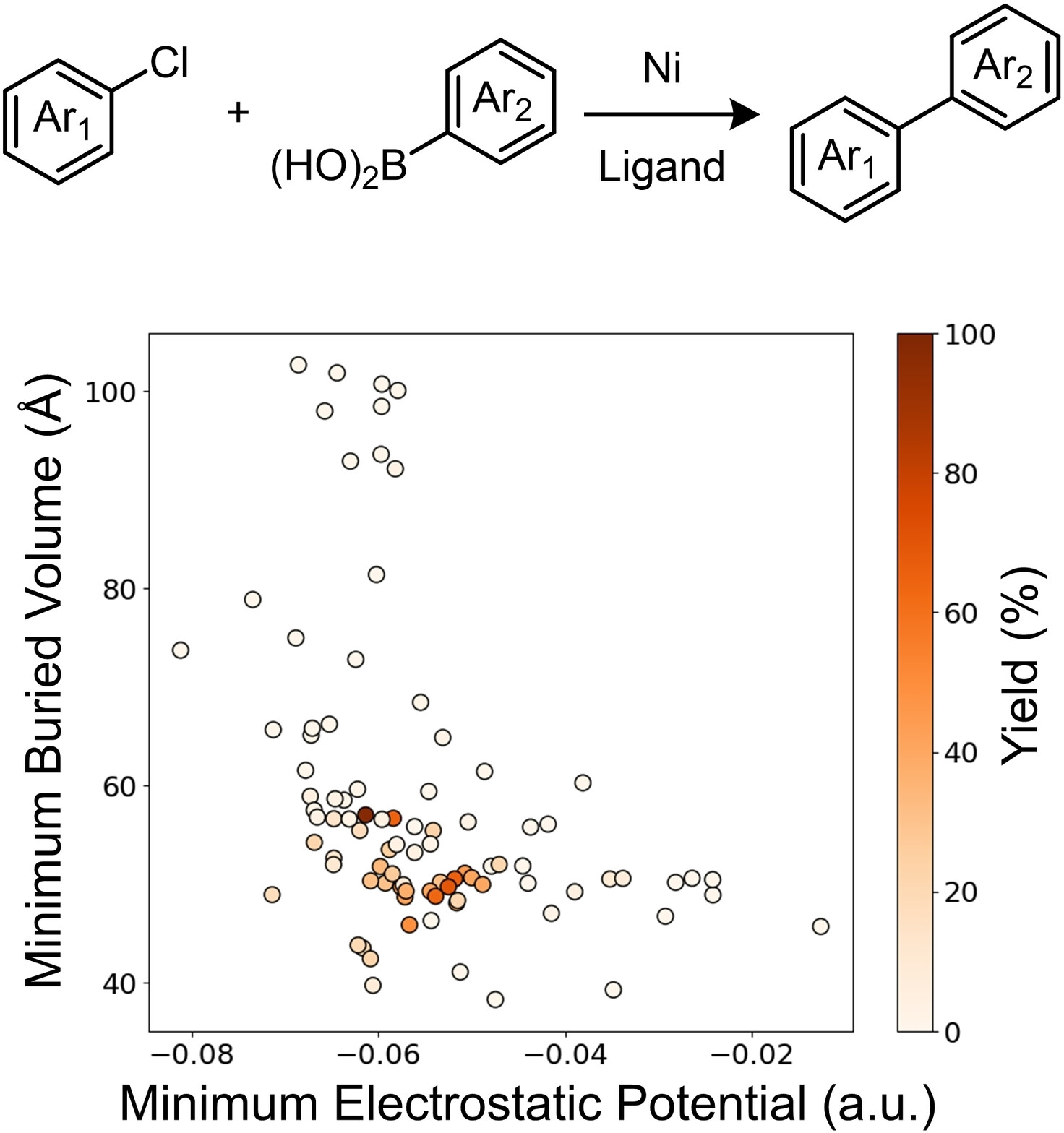
93. “Multi-Threshold Analysis for Chemical Space Mapping of Ni-Catalyzed Suzuki-Miyaura Couplings” LeSueur, A.; Tao, N.; Doyle, A.G.; Sigman, M. Eur. J. Org. Chem., 2024, e202400428 [DOI: 10.1002/ejoc.202400428] Link PDF
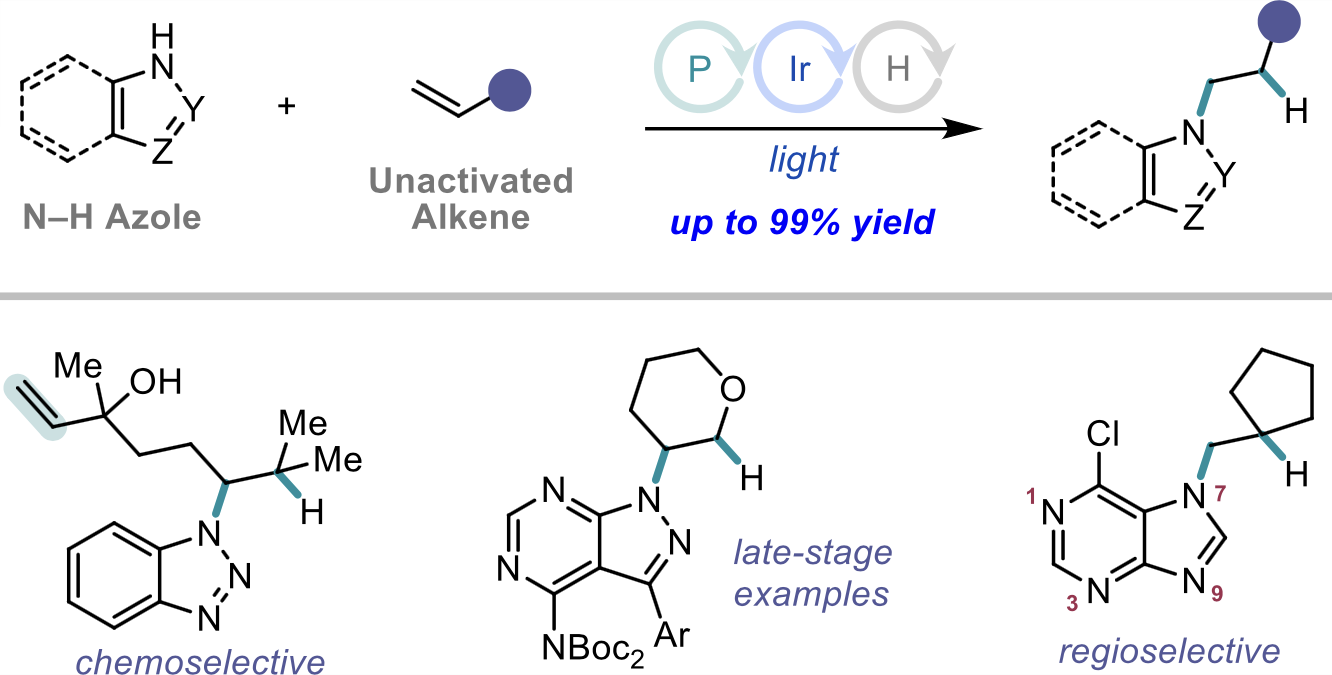
92. “Cooperative Phosphine-Photoredox Catalysis Enables N–H Activation of Azoles for Intermolecular Olefin Hydroamination” Sedillo, K.; Fan, F.; Knowles, R. R.; Doyle, A. G. J. Am. Chem. Soc. 2024, 146, 20349-20356. [DOI: 10.1021/jacs.4c05881] Link PDF
91. “Mechanism of Ni-Catalyzed Photochemical Halogen Atom-Mediated C(sp3)–H Arylation” Cusumano, A. Q; Chaffin, B. C.; Doyle, A. G. J. Am. Chem. Soc. 2024, 146, 15331-15344. [DOI: 10.1021/jacs.4c03099] Link PDF
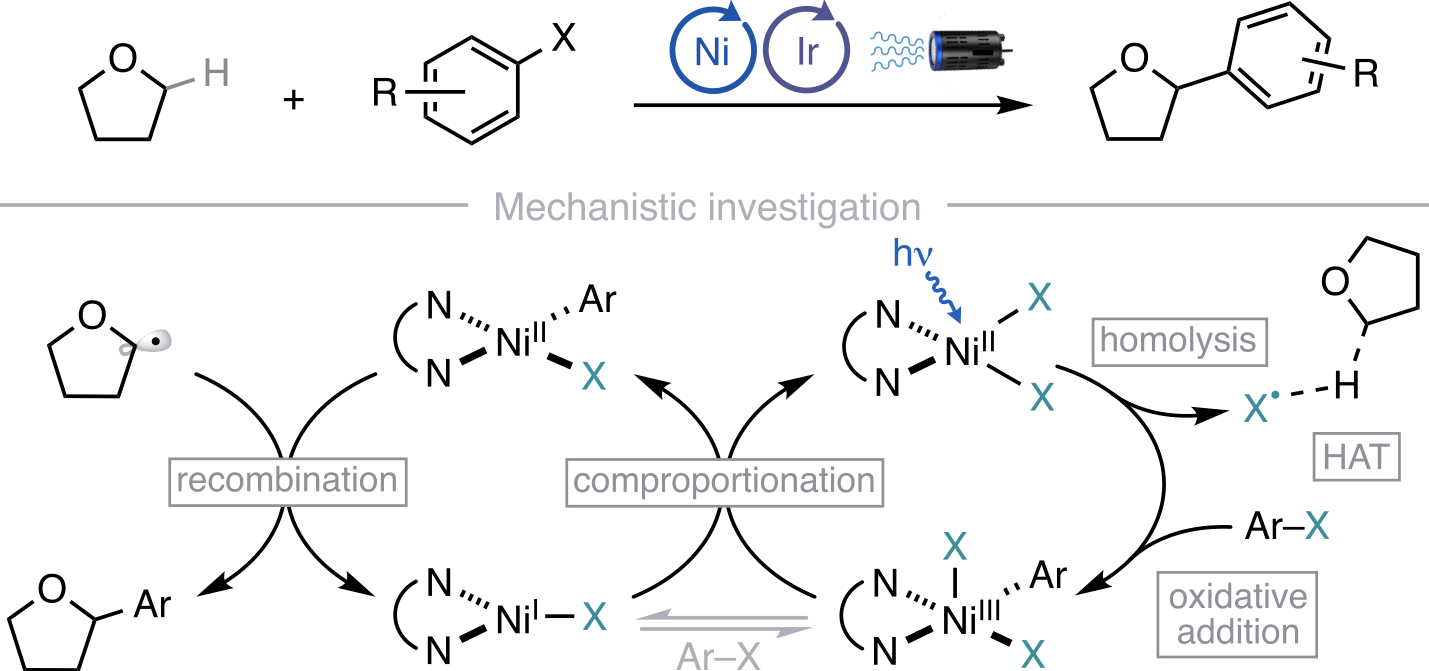
90. “Autonomous closed-loop mechanistic investigation of molecular electrochemistry via automation” Sheng, H.; Sun, J.; Rodríguez, O.; Hoar, B. B.; Zhang, W.; Xiang, D.; Tang, T.; Hazra, A.; Min, D. S.; Doyle, A. G.; Sigman, M. S.; Costentin, C.; Gu, Q.; Rodríguez-López, J.; Liu, C. Nat. Commun. 2024, 15, 2781. [DOI: 10.1038/s41467-024-47210-x] Link PDF

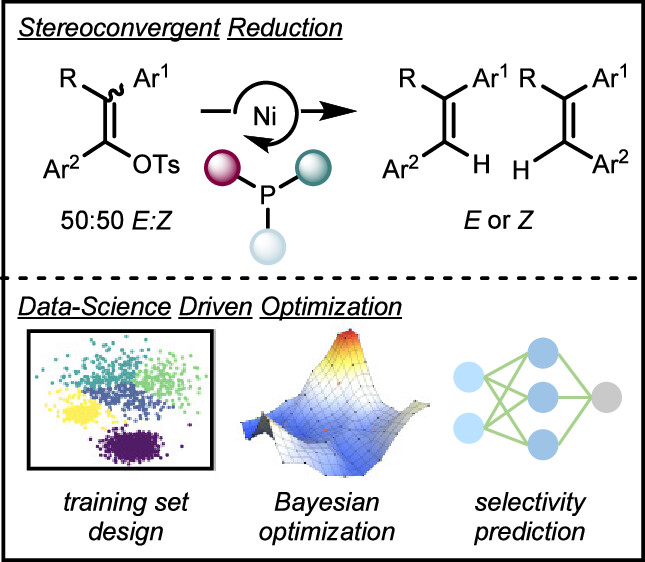
89. “Data Science Guided Multiobjective Optimization of a Stereoconvergent Nickel-Catalyzed Reduction of Enol Tosylates to Access Trisubstituted Alkenes” Romer, N. P.; Min, D. S.; Wang, J. Y.; Walroth, R. C.; Mack, K. A.; Sirois, L. E.; Gosselin, F.; Zell, D.; Doyle, A. G.; Sigman, M. S. ACS Catal, 2024, 14, 4699-4708. [DOI: 10.1021/acscatal.4c00650] Link PDF
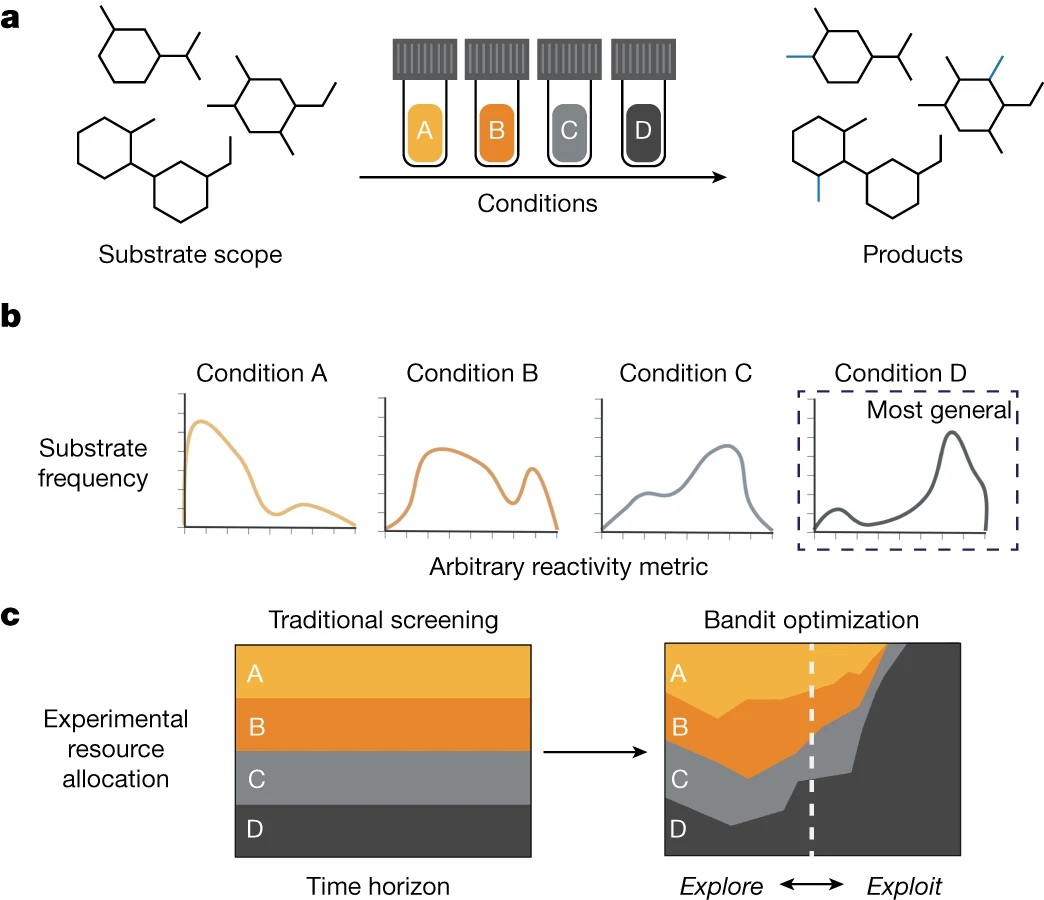
88. “Identifying general reaction conditions by bandit optimization” Wang, J. Y.; Stevens, J. M.; Kariofillis, S. K.; Tom, M.-J.; Golden, D. L.; Li, J.; Tabora, J. E.; Parasram, M.; Shields, B. J.; Primer, D. N.; Hao, B.; Del Valle, D.; DiSomma, S.; Furman, A.; Zipp, G. G.; Melnikov, S.; Paulson, J.; Doyle, A. G. Nature, 2024, 626, 1025-1033. [DOI: 10.1038/s41586-024-07021-y] Link PDF
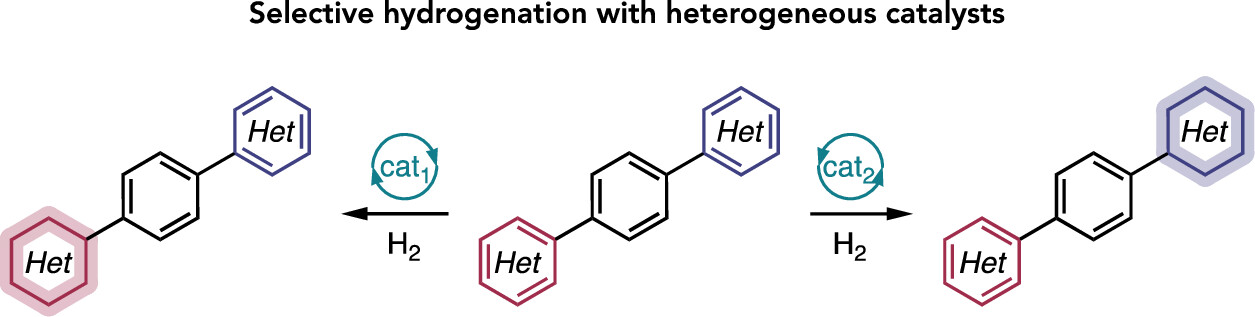
87. “Broad Survey of Selectivity in the Heterogeneous Hydrogenation of Heterocycles” Lyons, T. W.; Leibler, I. N.-M.; He, C. Q.; Gadamsetty, S.; Estrada, G. J.; Doyle, A. G. J. Org. Chem. 2024, 89, 1438-1445. [DOI: 10.1021/acs.joc.3c02028] Link PDF
2023
86. “Catalyst Deactivation of a Monoligated CyJohnPhos-Bound Nickel(0) Complex” Newman-Stonebraker, S. H.; Raab, T. J.; Doyle, A. G. Organometallics 2023, 42, 3438-3441. [DOI: 10.1021/acs.organomet.3c00450] Link PDF
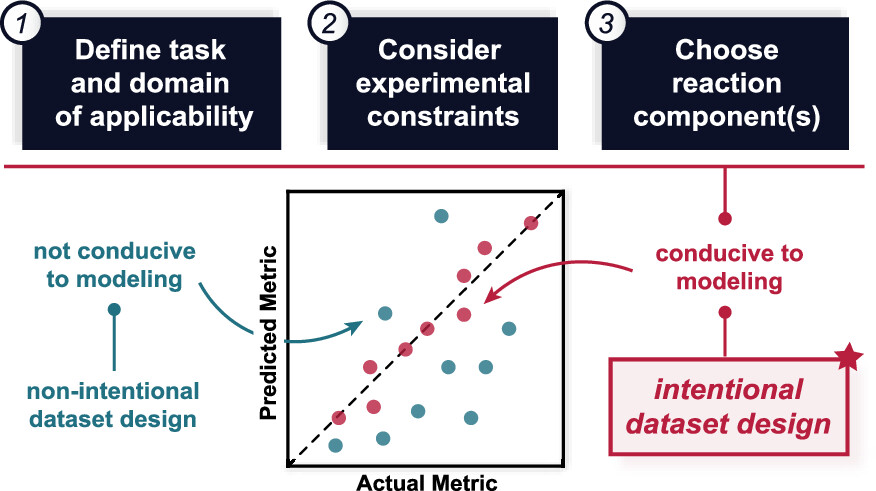
85. “Dataset Design for Building Models of Chemical Reactivity” Raghavan, P; Haas, B. C.; Ruos, M. E.; Schleinitz, J; Doyle, A. G.; Reisman, S. E.; Sigman, M. S.; Coley, C. W. ACS Cent. Sci. 2023, 9, 2196-2204. [DOI: 10.1021/acscentsci.3c01163] Link PDF
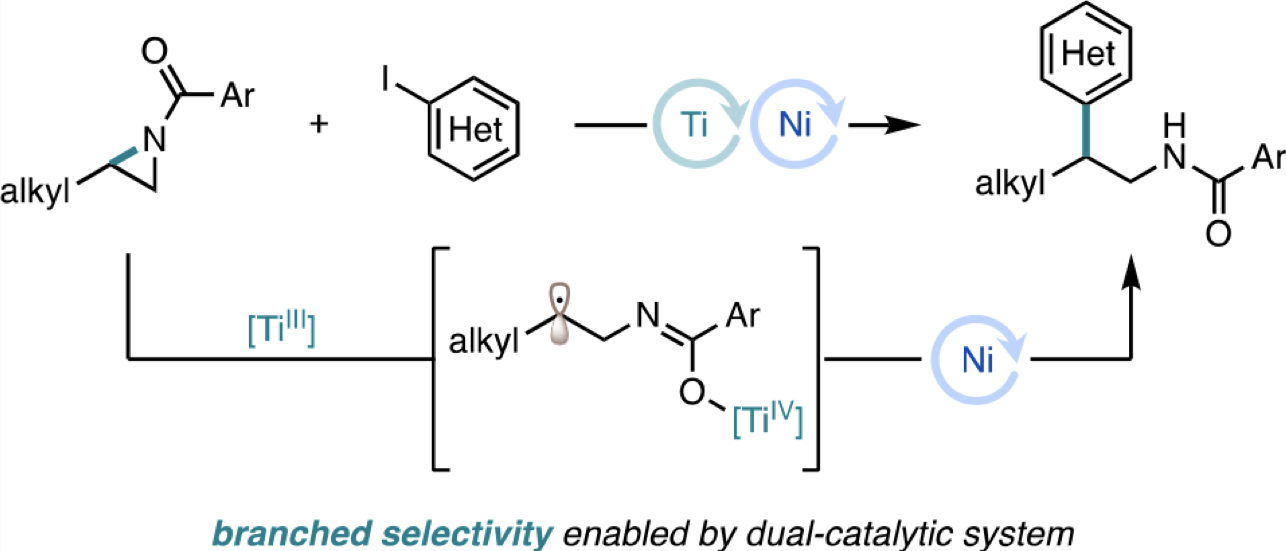
84. “Branched-Selective Cross-Electrophile Coupling of 2-Alkyl Aziridines and (Hetero)aryl Iodides Using Ti/Ni Catalysis” Williams, W. L.; Gutiérrez-Valencia, N. E.; Doyle, A. G. J. Am. Chem. Soc. 2023, 145, 24175-24183. [DOI: 10.1021/jacs.3c08301] Link PDF
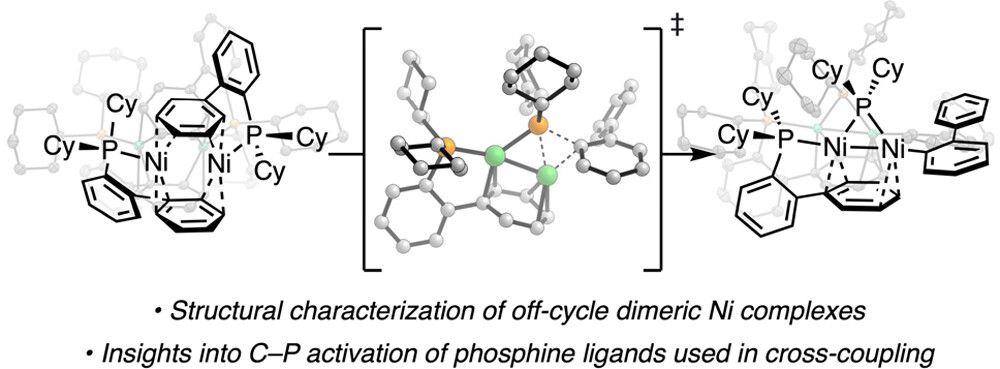
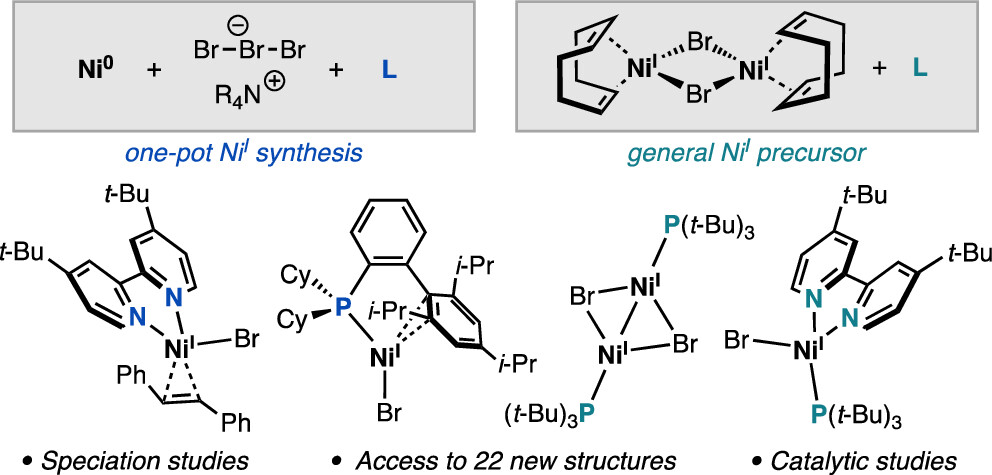
83. “Synthesis of Nickel(I)–Bromide Complexes via Oxidation and Ligand Displacement: Evaluation of Ligand Effects on Speciation and Reactivity” Newman-Stonebraker, S. H.; Raab, T. J.; Roshandel, H. R.; Doyle, A. G. J. Am. Chem. Soc. 2023, 145, 19368-19377. [DOI: 10.1021/jacs.3c06233] Link PDF
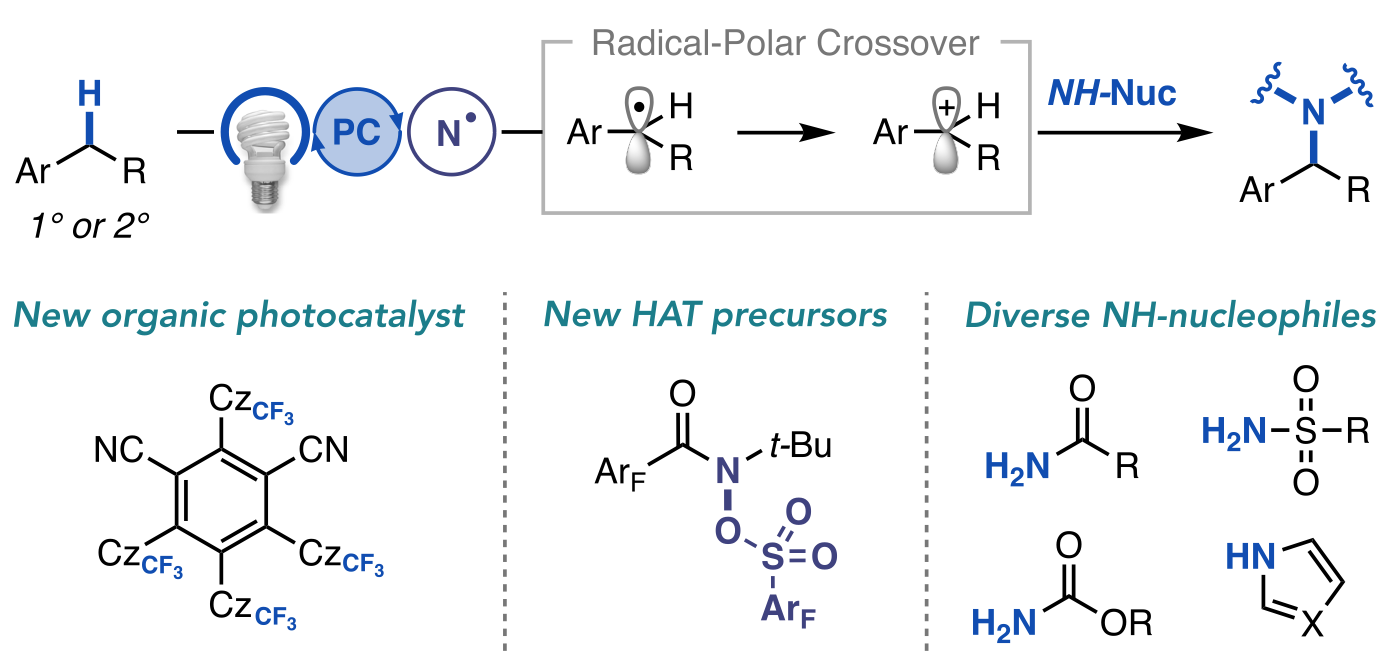
82. “A General Photocatalytic Strategy for Nucleophilic Amination of Primary and Secondary Benzylic C–H Bonds” Ruos, M. E.; Kinney, R. G.; Ring, O. T.; Doyle, A. G. J. Am. Chem. Soc. 2023, 145, 18487-18496. [DOI: 10.1021/jacs.3c04912] Link PDF
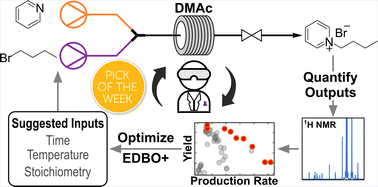
81. “Continuous flow synthesis of pyridinium salts accelerated by multi-objective Bayesian optimization with active learning” Dunlap, J. H.; Ethier, J. G.; Putnam-Neeb, A. A.; Iyer, S.; Luo, S.-X. L.; Feng, H.; Torres, J. A. G.; Doyle, A. G.; Swager, T. M.; Vaia, R. A.; Mirau, P.; Crouse, C. A.; Baldwin, L. A. Chem. Sci. 2023, 14, 8061-8069. [DOI: 10.1039/d3sc01303k] Link PDF
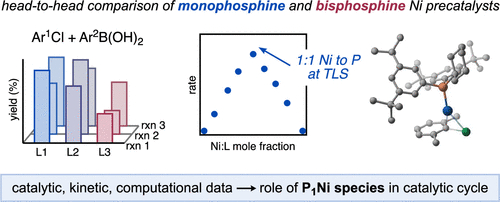
80. “Comparison of Monophosphine and Bisphosphine Precatalysts for Ni-Catalyzed Suzuki–Miyaura Cross-Coupling: Understanding the Role of the Ligation State in Catalysis” Borowski, J. E.; Newman-Stonebraker, S. H.; Doyle, A. G. ACS. Catal. 2023, 13, 7966-7977. [DOI: 10.1021/acscatal.3c01331] Link PDF
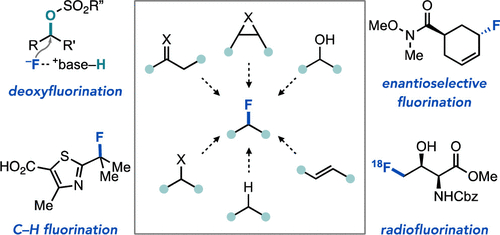
79. “Strategies for Nucleophilic C(sp3)–(Radio)Fluorination” Leibler, I. N.-M.; Gandhi, S. S.; Tekle-Smith, M. A.; Doyle, A. G. J. Am. Chem. Soc. 2023, 145, 9928-9950. [DOI: 10.1021/jacs.3c01824] Link PDF
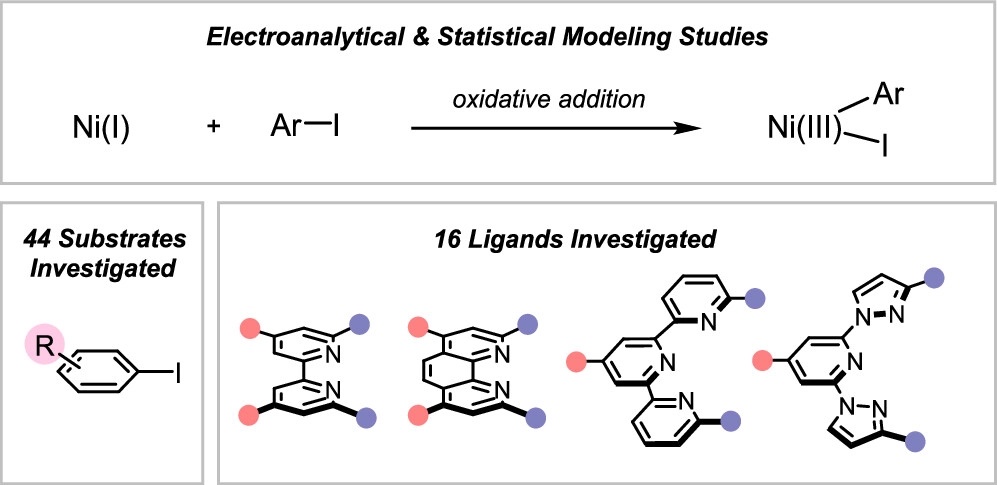
78. “Interrogating the Mechanistic Features of Ni(I)-Mediated Aryl Iodide Oxidative Addition Using Electroanalytical and Statistical Modeling Techniques” Tang, T.; Hazra, A.; Min, D. S.; Williams, W. L.; Doyle, A. G.; Sigman, M. S. J. Am. Chem. Soc. 2023, 145, 8689-8699. [DOI: 10.1021/jacs.3c01726] Link PDF
77. “A Machine Learning Approach to Model Interaction Effects: Development and Application to Alcohol Deoxyfluorination” Żurański, A. M.; Gandhi, S. S.; Doyle, A. G. J. Am. Chem. Soc. 2023, 145, 7898–7909. [DOI: 10.1021/jacs.2c13093] Link PDF
76. “On the Use of Real-World Datasets for Reaction Yield Prediction” Saebi, M.; Nan, B.; Herr, J. E.; Wahlers, J.; Guo, Z.; Zurański, A. M.; Kogej, T.; Norrby, P.-O.; Doyle, A. G.; Wiest, O.; Chawla, N. V. Chem. Sci. 2023 [DOI: 10.1039/d2sc06041h] Link PDF
2022
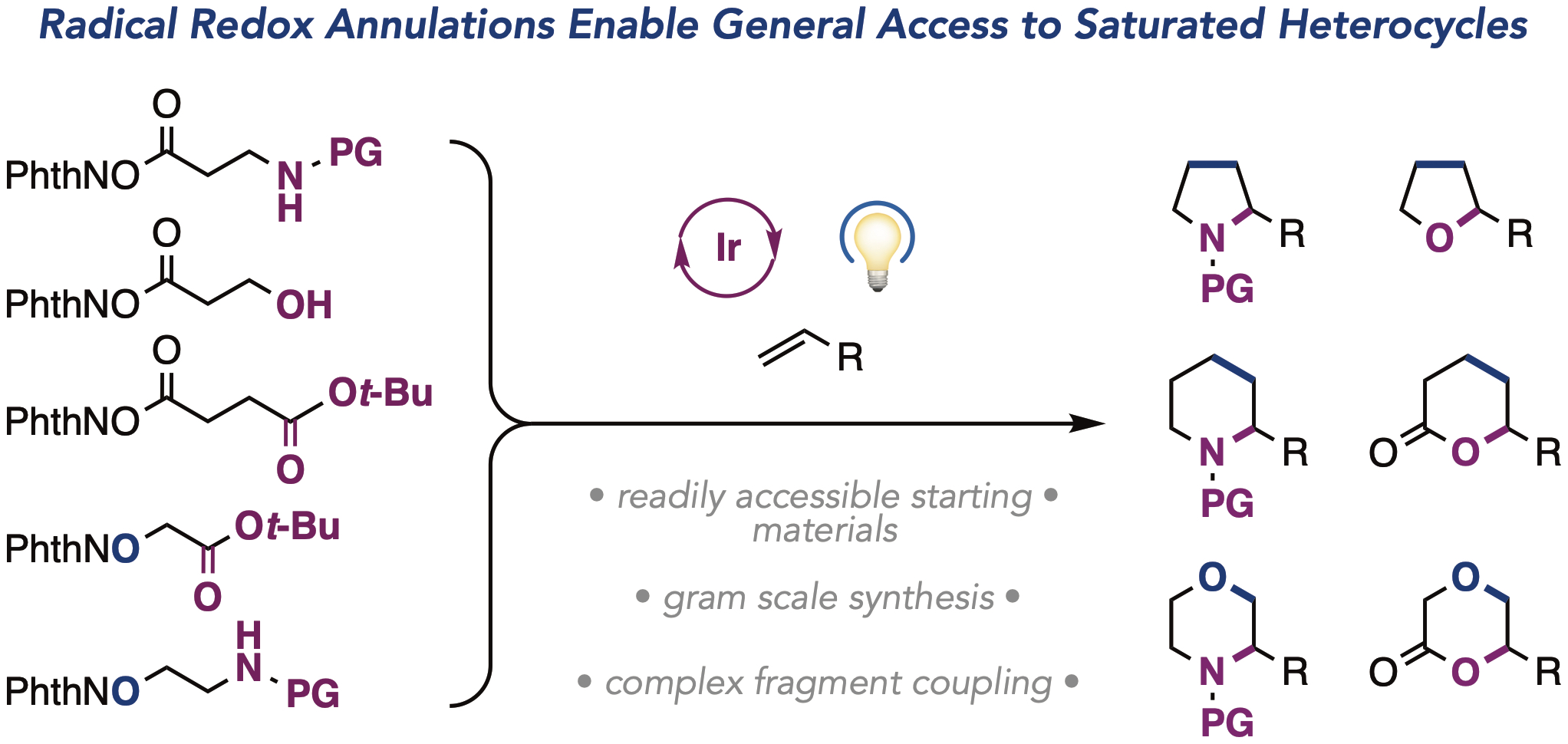
75. “Radical Redox Annulations: A General Light-Driven Method for the Synthesis of Saturated Heterocycles” Murray, P. R. D.; Leibler, I. N.-M.; Hell, S. M.; Villalona, E.; Doyle, A. G.; Knowles, R. R. ACS Catal. 2022, 12, 13732-13740. [DOI: 10.1021/acscatal.2c04316] Link PDF
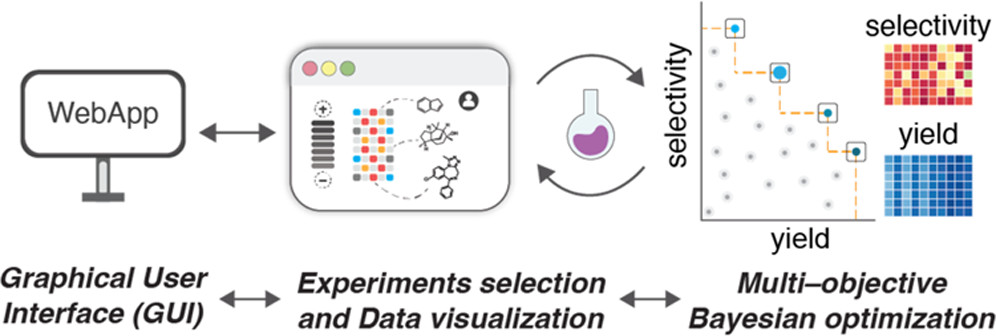
74. “A Multi-Objective Active Learning Platform and Web App for Reaction Optimization” Torres, J. A. G.; Lau, S. H.; Anchuri, P.; Stevens, J. M.; Tabora, J. E.; Li, J.; Borovika, A.; Adams, R. P.; Doyle, A. G. J. Am. Chem. Soc. 2022, 144, 19999–20007. [DOI: 10.1021/jacs.2c08592] Link PDF Link EDBO+ web app.
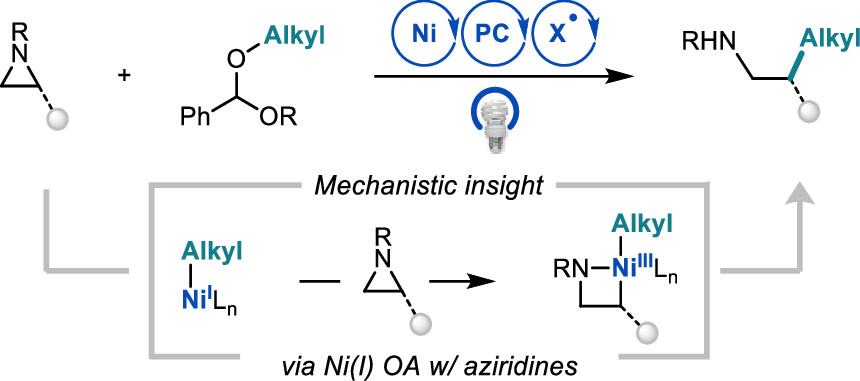
73. “Ni/Photoredox-Catalyzed C(sp3)–C(sp3) Coupling between Aziridines and Acetals as Alcohol-Derived Alkyl Radical Precursors” Dongbang, S; Doyle, A. G. J. Am. Chem. Soc. 2022, 144, 20067–20077. [DOI: 10.1021/jacs.2c09294] Link PDF
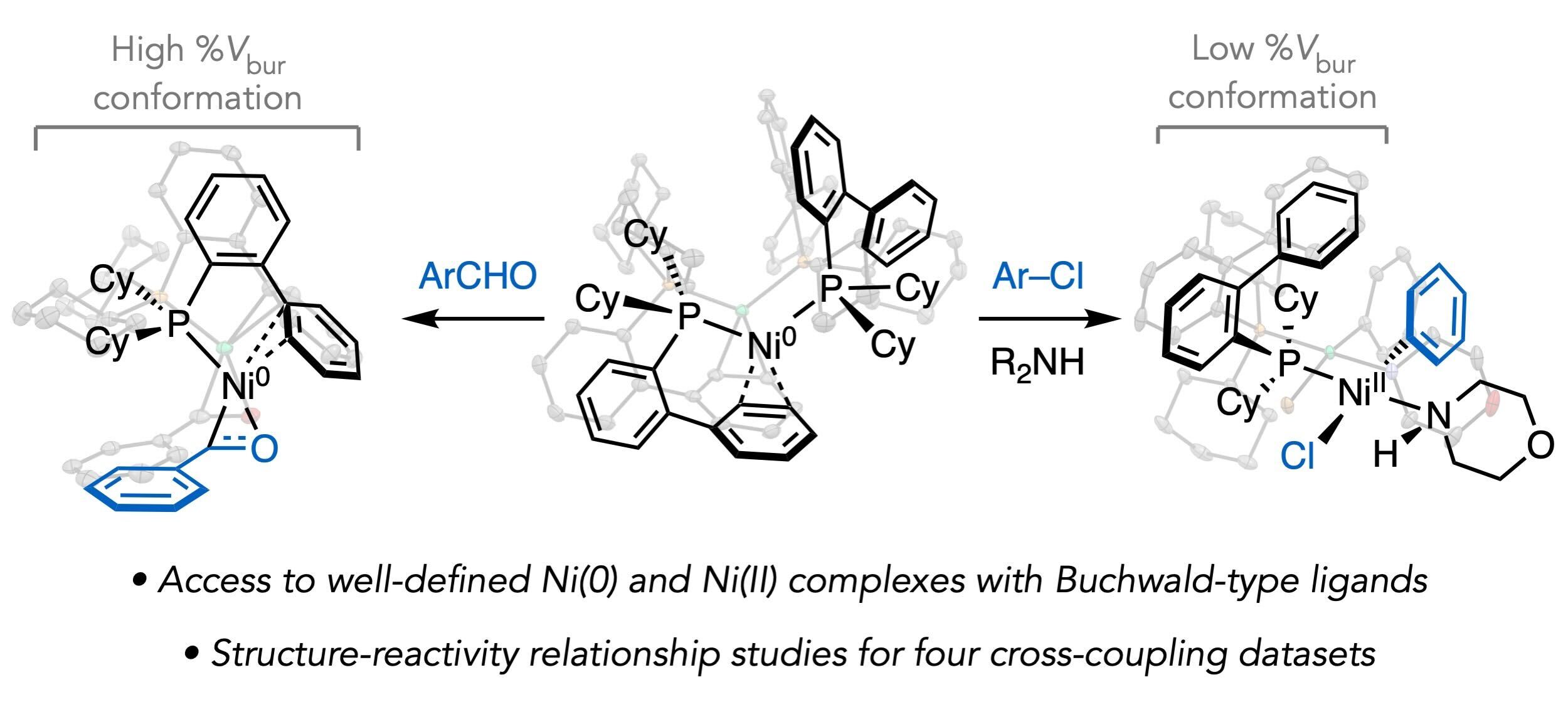
72. “Structure–Reactivity Relationships of Buchwald-Type Phosphines in Nickel-Catalyzed Cross-Couplings” Newman-Stonebraker, S. H.; Wang, J. Y.; Jeffrey P. D.; Doyle, A. G. J. Am. Chem. Soc. 2022, 144, 19635-19648. [DOI: 10.1021/jacs.2c09840] Link PDF
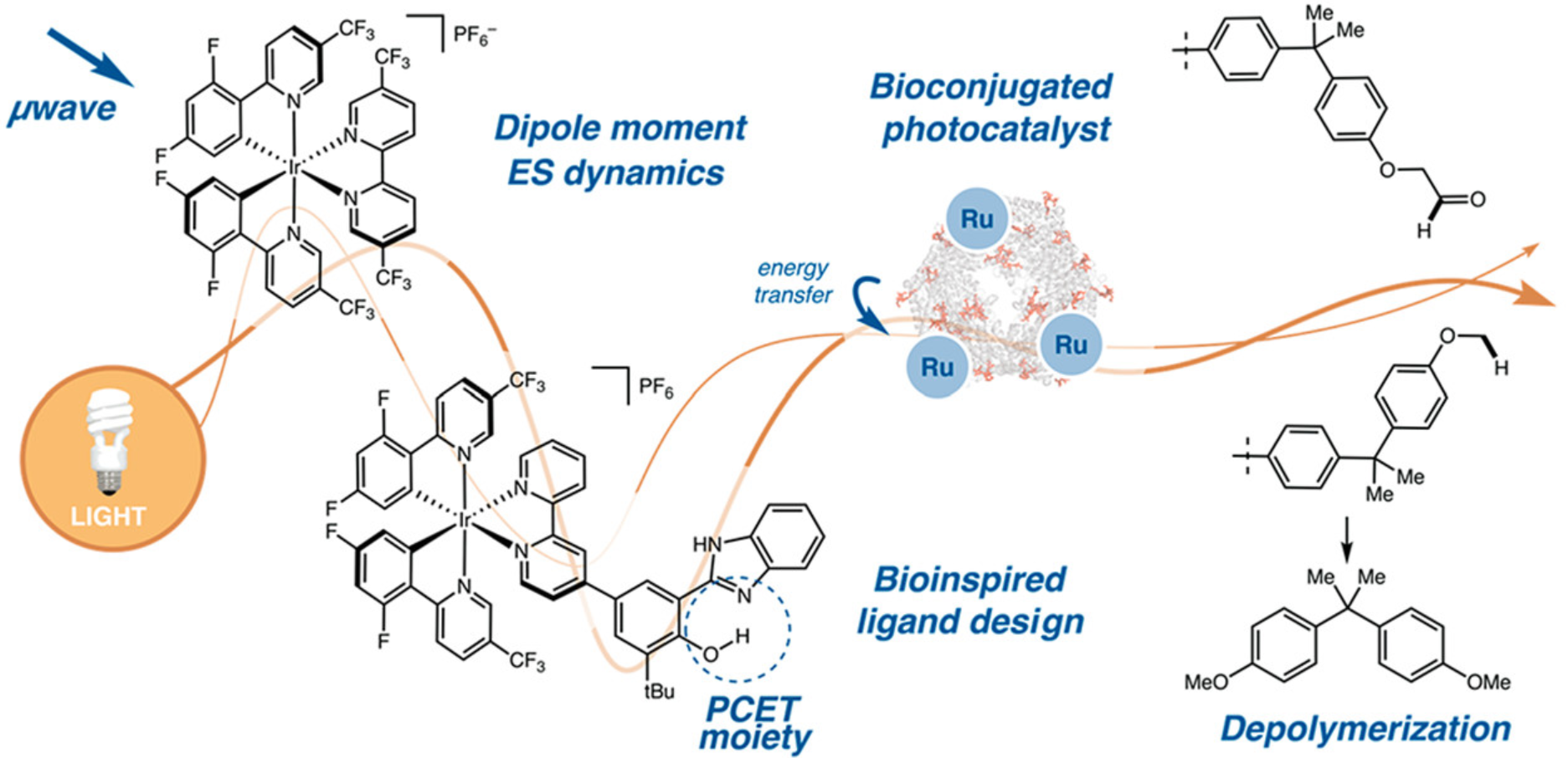
71. “Bioinspired Supercharging of Photoredox Catalysis for Applications in Energy and Chemical Manufacturing.” Millet, A.; Cesana, P. T.; Sedillo, K.; Bird, M. J.; Schlau-Cohen, G. S.; Doyle, A. G.; MacMillan, D. W. C.; Scholes, G. D. Acc. Chem. Res. 2022, 55, 1423-1434. [DOI: 10.1021/acs.accounts.2c00083] Link PDF
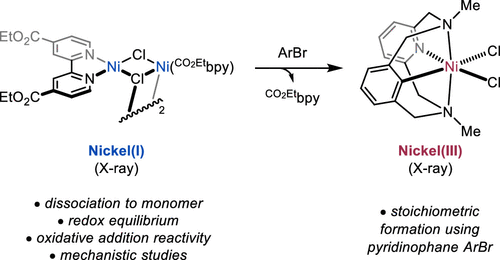
70. “Oxidative Addition of Aryl Halides to a Ni(I)-Bipyridine Complex.” Ting, S. I.; Williams, W. L.; Doyle, A. G. J. Am. Chem. Soc. 2022, 144, 5575-5582. [DOI: 10.1021/jacs.2c00462] Link PDF
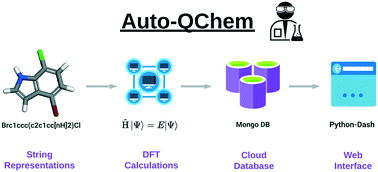
69. “Auto-QChem: an automated workflow for the generation and storage of DFT calculations for organic molecules.” Żurański, A. M.; Wang, J. Y.; Shields, B. J.; Doyle, A. G. React. Chem. Eng. 2022, 7, 1276-1284. [DOI: 10.1039/D2RE00030J] Link PDF
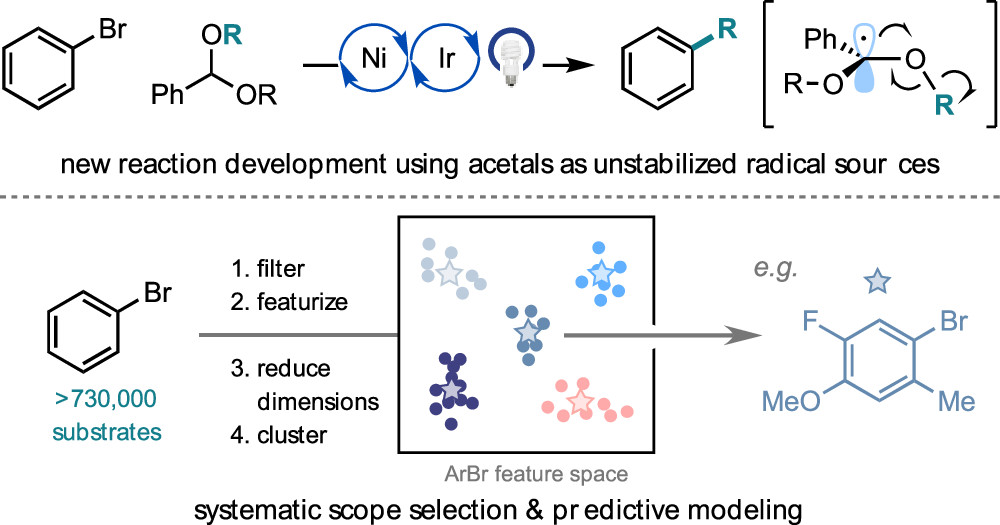
68. “Using Data Science to Guide Aryl Bromide Substrate Scope Analysis in a Ni/Photoredox-Catalyzed Cross-Coupling with Acetals as Alcohol-Derived Radical Sources.” Kariofillis, S. K.; Jiang, S.; Żurański, A. M.; Gandhi, S. S.; Martinez Alvarado, J. I.; Doyle, A. G. J. Am. Chem. Soc. 2022, 144, 1045-1055. [DOI: 10.1021/jacs.1c12203] Link PDF
2021
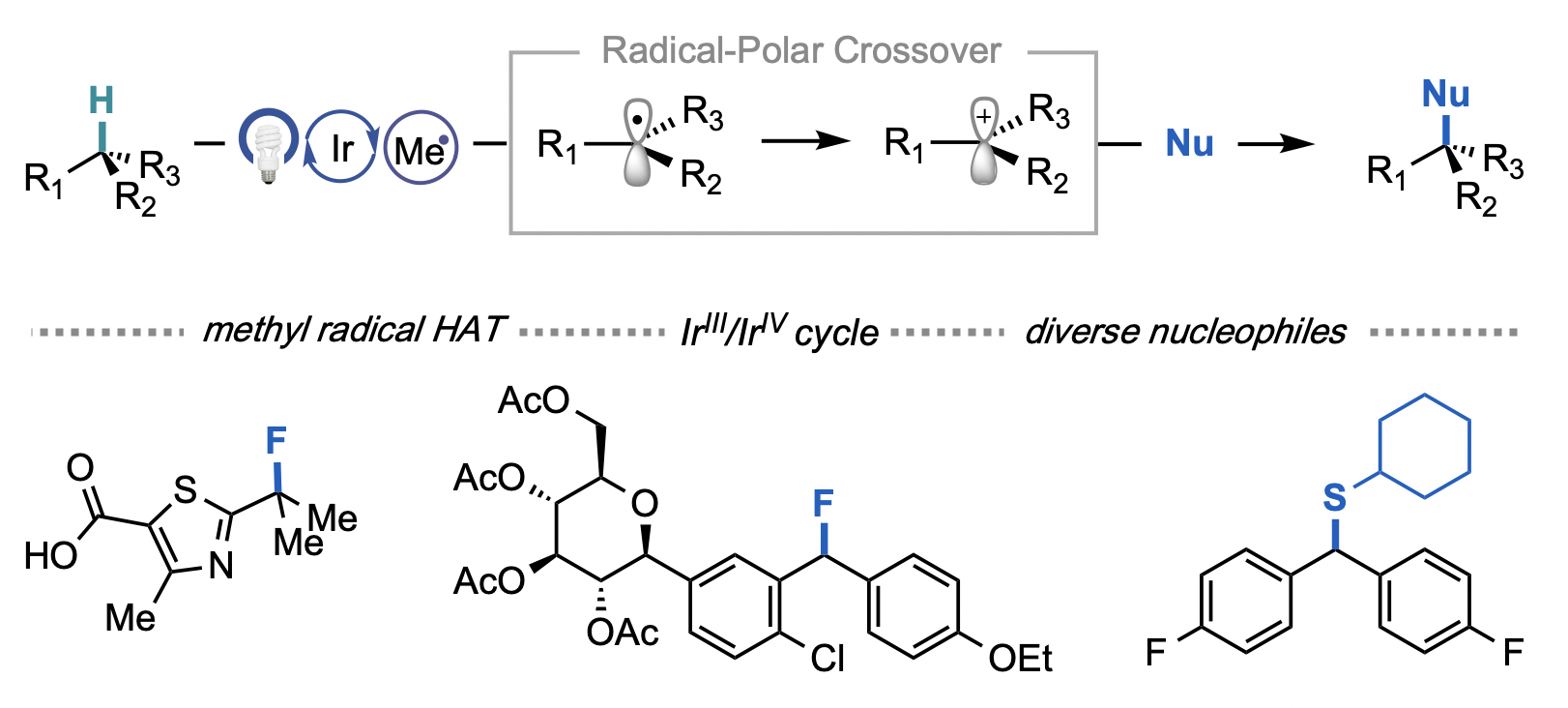
67. “A General Strategy for C(sp3)–H Functionalization with Nucleophiles Using Methyl Radical as a Hydrogen Atom Abstractor.” Leibler, I. N.-M.; Tekle-Smith, M. A.; Doyle, A. G. Nat. Commun. 2021, 12, 6950. [DOI: 10.1038/s41467-021-27165-z] Link PDF
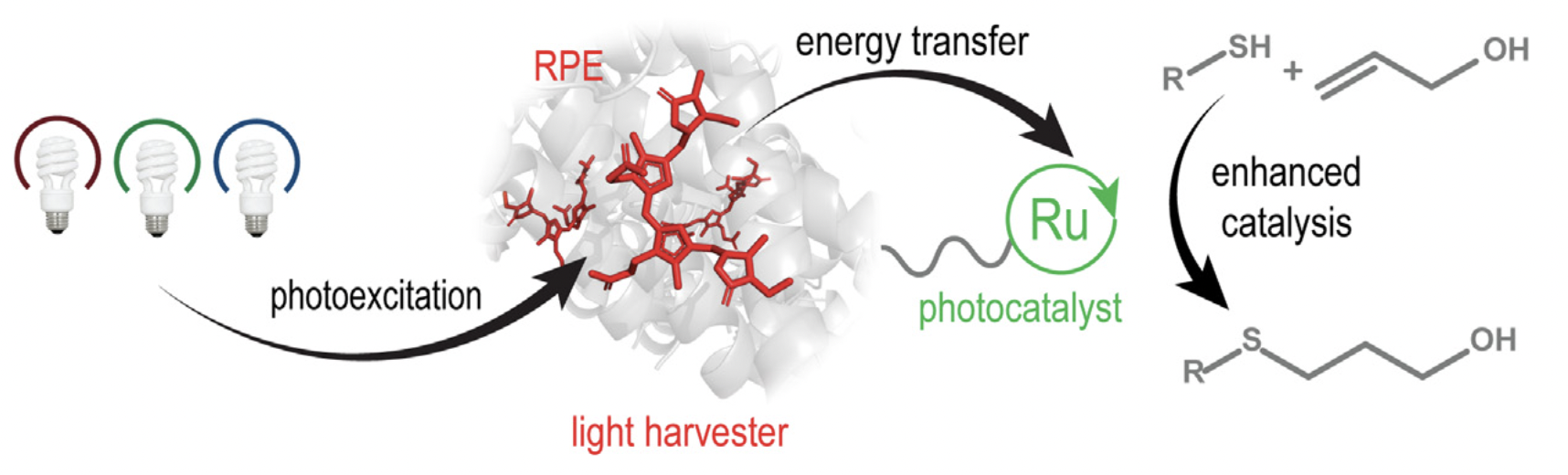
66. “A Biohybrid Strategy for Enabling Photoredox Catalysis with Low-Energy Light.” Cesana, P. T.; Li, B. X.; Shepard, S. G.; Ting, S. I.; Hart, S. M.; Olson, C. M.; Martinez Alvarado, J. I.; Son, M.; Steiman, T. J.; Castellano, F. N.; Doyle, A. G.; MacMillan, D. W. C.; Schlau-Cohen, G. S. Chem. 2021, 8, 174-185. [DOI: 10.1016/j.chempr.2021.10.010] Link PDF
65. “The Open Reaction Database.” Kearnes, S. M.; Maser, M. R.; Wleklinski, M.; Kast, A.; Doyle, A. G.; Dreher, S. D.; Hawkins, J. M.; Jensen, K. F.; Coley, C. W. J. Am. Chem. Soc. 2021, 143, 18820-18826. [DOI: 10.1021/jacs.1c09820] Link PDF
64. “The Evolution of Data-Driven Modeling in Organic Chemistry.” Williams, W. L.; Zeng, L.; Gensch, T.; Sigman, M. S.; Doyle, A. G.; Anslyn, E. V. ACS Cent. Sci. 2021, 7, 1622-1637. [10.1021/acscentsci.1c00535] Link PDF
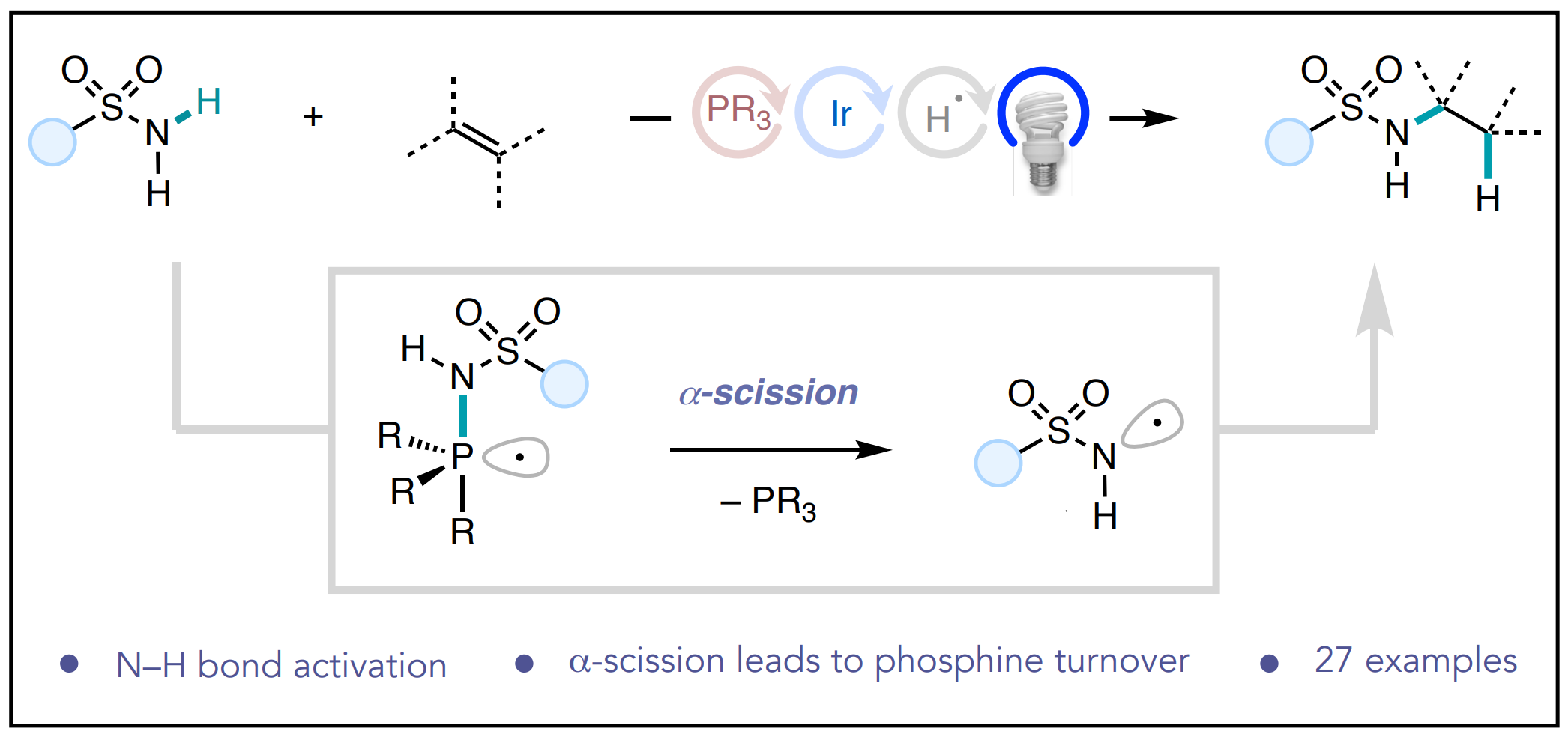
63. “Phosphine/Photoredox Catalyzed Anti-Markovnikov Hydroamination of Olefins with Primary Sulfonamides via α-Scission from Phosphoranyl Radicals.” Chinn, A. J.; Sedillo, K.; Doyle, A. G. J. Am. Chem. Soc. 2021, 143, 18331-18338. [DOI: 10.1021/jacs.1c09484] Link PDF
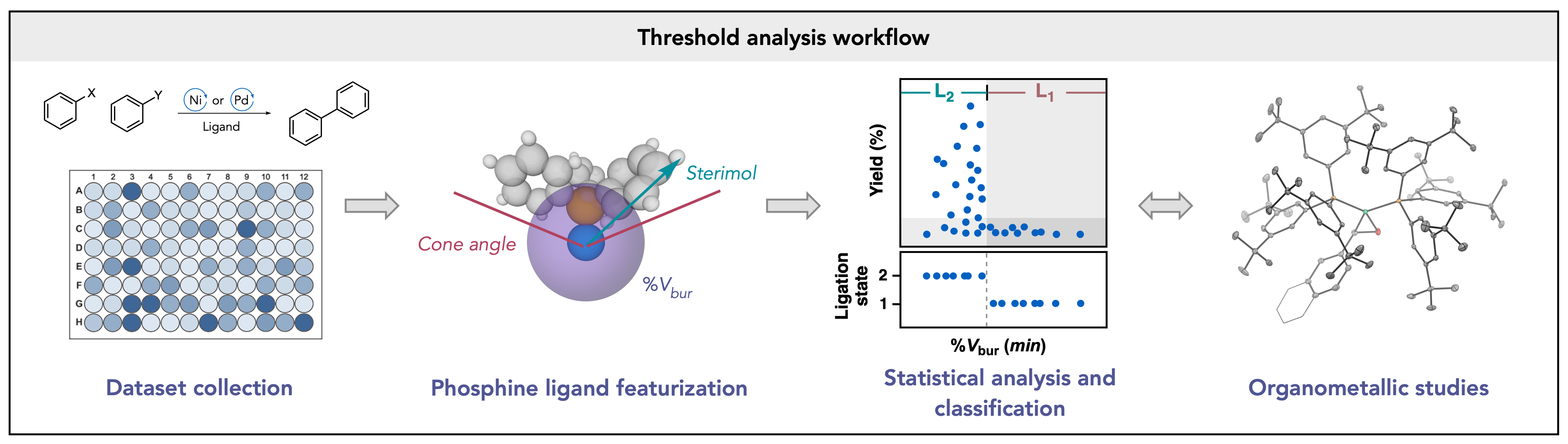
62. “Univariate classification of phosphine ligation state and reactivity in cross-coupling catalysis.” Newman-Stonebraker, S. H.; Smith, S. R.; Borowski, J. E.; Peters, E.; Gensch, T.; Johnson, H. C.; Sigman, M. S.; Doyle, A. G. Science 2021, 374, 301-308. [DOI: 10.1126/science.ajb4213] Link PDF
61. “Ni/Photoredox-Catalyzed Enantioselective Cross-Electrophile Coupling of Styrene Oxides with Aryl Iodides.” Lau, S. H.; Borden, M. A.; Steiman, T. J.; Parasram, M.; Wang, L. S.; Doyle, A. G. J. Am. Chem. Soc 2021, 143, 15873-15881. [DOI: 10.1021/jacs.1c08105] Link PDF
60. “Predicting Reaction Yields via Supervised Learning.” Żurański, A. M.; Martinez Alvarado, J. I.; Shields, B. J.; Doyle, A. G. Acc. Chem. Res 2021, 54, 1856-1865. [DOI: 10.1021/acs.accounts.0c00770] Link PDF
59. “Automation and computer-assisted planning for chemical synthesis.” Shen, Y.; Borowski, J. E.; Hardy, M. A.; Sarpong, R.; Doyle, A. G.; Cernak, T. Nat. Rev. Methods Primers 2021, 1, 23. [DOI: 10.1038/s43586-021-00022-5] Link PDF
58. “Bayesian reaction optimization as a tool for chemical synthesis.” Shields, B. J.; Stevens, J.; Li, J.; Parasram, M.; Damani, F.; Martinez Alvarado, J. I.; Janey, J. M.; Adams, R. P.; Doyle, A. G. Nature 2021, 590, 89-96. [DOI: 10.1038/s41586-021-03213-y] Link PDF Github
57. “Synthetic and Mechanistic Implications of Chlorine Photoelimination in Nickel/Photoredox C(sp3)–H Cross-Coupling.” Kariofillis, S. K.; Doyle, A. G. Acc. Chem. Res. 2021, 54, 988-1000. [DOI: 10.1021/acs.accounts.0c00694] Link PDF
2020
56. “Bioinspiration in Light Harvesting and Catalysis.” Proppe, A. H.; Li, Y. C.; Aspuru-Guzik, A.; Berlinguette, C. P.; Chang, C. J.; Cogdell, R.; Doyle, A. G.; Flick, J.; Gabor, N. M.; van Grondelle, R.; Hammes-Schiffer, S.; Jaffer, S. A.; Kelley, S. O.; Leclerc, M.; Leo, K.; Mallouk, T. E.; Narang, P.; Schlau-Cohen, G. S.; Scholes, G. D.; Vojvodic, A.; Yam, V. W.; Yang, J. Y.; Sargent, E. H. Nat. Rev. Mater. 2020, 5, 828-846. [DOI: 10.1038/s41578-020-0222-0] Link PDF
55. “Nucleophilic (Radio)Fluorination of Redox-Active Esters via Radical-Polar Crossover Enabled by Photoredox Catalysis.” Webb, E. W.; Park, J. B.; Cole, E. L.; Donnelly, D. J.; Bonacorsi, S. J.; Ewing, W. R.; Doyle, A. G. J. Am. Chem. Soc. 2020, 142, 9493-9500. [DOI: 10.1021/jacs.0c03125] Link PDF

54. “Regioselective Cross-Electrophile Coupling of Epoxides and (Hetero)aryl Iodides via Ni/Ti/Photoredox Catalysis.” Parasram, M.; Shields, B. J.; Ahmad, O.; Knauber, T.; Doyle, A. G. ACS Catal 2020, 10, 5821-5827. [DOI: 10.1021/acscatal.0c01199] Link PDF

53. “Role of Electron-Deficient Olefin Ligands in a Ni-Catalyzed Aziridine Cross-Coupling To Generate Quaternary Carbons.” Estrada, J. G.; Williams, W. L.; Ting, S. I.; Doyle, A. G. J. Am. Chem. Soc. 2020, 142, 8928-8937. [DOI: 10.1021/jacs.0c02237] Link PDF

52. “Nickel/Photoredox-Catalyzed Methylation of (Hetero)aryl Chlorides Using Trimethyl Orthoformate as a Methyl Radical Source.” Kariofillis, S. K.; Shields, B. J.; Tekle-Smith, M. A.; Zacuto, M. J.; Doyle, A. G. J. Am. Chem. Soc. 2020, 142, 7683-7689. [DOI: 10.1021/jacs.0c02805] Link PDF

51. “Synthesis of β-Phenethylamines via Ni/Photoredox Cross-Electrophile Coupling of Aliphatic Aziridines and Aryl Iodides.” Steiman, T. J.; Liu, J.; Mengiste, A.; Doyle, A. G. J. Am. Chem. Soc. 2020, 142, 7598-7605. [DOI: 10.1021/jacs.0c01724] Link PDF

50. “3d-d Excited States of Ni(II) Complexes Relevant to Photoredox Catalysis: Spectroscopic Identification and Mechanistic Implications.” Ting, S. I.; Garakyaraghi, S.; Taliaferro, C. M.; Shields, B. J.; Scholes, G. D.; Castellano, F. N.; Doyle, A. G. J. Am. Chem. Soc. 2020, 142, 5800-5810. [DOI: 10.1021/jacs.0c00781] Link PDF

2019
49. “Direct Use of Carboxylic Acids in the Photocatalytic Hydroacylation of Styrenes to Generate Dialkyl Ketones.” Martinez Alvarado, J. I.; Ertel, A. B.; Stegner, A.; Stache, E.; Doyle, A. G. Org. Lett. 2019, 21, 9940-9944. [DOI: 10.1021/acs.orglett.9b03871] Link PDF

2018
48. “Discrete Object Generation with Reversible Inductive Construction.” Seff, A.; Zhou, W.; Damani, F.; Doyle, A. G.; Adams, R. P. [DOI: arXiv:1907.08268 [cs.LG]] Link PDF
47. “Response to Comment on ‘Predicting Reaction Performance in C-N Cross-Coupling Using Machine Learning.” Estrada, J. G.; Ahneman, D. T.; Sheridan, R. P.; Dreher, S. D.; Doyle, A. G. Science 2018, 362, 17402–17404. [10.1126/science.aat8763] Link PDF Github

46. “Generation of Phosphoranyl Radicals via Photoredox Catalysis Enables Voltage-Independent Activation of Strong C-O Bonds.” Stache, E. E.; Ertel, A. B.; Rovis, T.; Doyle, A. G. ACS Catal. 2018, 8, 11134-11139. [DOI: 10.1021/acscatal.8b03592] Link PDF

45. “Direct C-C Bond Formation from Alkanes Using Ni-Photoredox Catalysis.” Ackerman, L. K. G.; Martinez Alvarado, J. I.; Doyle, A. G. J. Am. Chem. Soc. 2018, 140, 14059-14063. [DOI: 10.1021/jacs.8b09191] Link PDF

44. “Deoxyfluorination with Sulfonyl Fluorides: Navigating Reaction Space with Machine Learning.” Nielsen, M. K.; Ahneman, D. T.; Riera, O.; Doyle, A. G. J. Am. Chem. Soc. 2018, 140, 5004-5008. [DOI: 10.1021/jacs.8b01523] Link PDF

43. “Predicting Reaction Performance in C-N Cross-Coupling Using Machine Learning.” Ahneman, D. T.; Estrada, J. G.; Lin, S.; Dreher, S. D.; Doyle, A. G. Science 2018, 360, 186-190. [DOI: 10.1126/science.aar5169] Link PDF

42. “Long-Lived Charge Transfer States of Nickel(II) Aryl Halide Complexes Facilitate Bimolecular Photoinduced Electron Transfer.” Shields, B. J.; Kudisch, B.; Scholes, G. D.; Doyle, A. G. J. Am. Chem. Soc. 2018, 140, 3035-3039. [DOI: 10.1021/jacs.7b13281] Link PDF

41. “Ni-Catalyzed Carbon-Carbon Bond-Forming Reductive Amination.” Heinz, C.; Lutz, J. P.; Simmons, E. M.; Miller, M. M.; Ewing, W. R.; Doyle, A. G. J. Am. Chem. Soc. 2018, 140, 2292-2300. [10.1021/jacs.0c03125] Link PDF

2017
40. “Mild, Redox-Neutral Formylation of Aryl Chlorides through the Photocatalytic Generation of Chlorine radicals.” Nielsen, M. K.; Shields, B. J.; Liu, J. Williams, M. J.; Zacuto, M. J; Doyle, A. G. Angew. Chem. Int. Ed. 2017, 56, 7191-7194. [DOI: 10.1002/ange.201702079] Link PDF

39. “Nickel-Catalyzed Enantioselective Reductive Cross-Coupling of Styrenyl Aziridines.” Woods, B. P.; Orlandi, M.; Huang, C.-Y. Sigman, M. H.; Doyle, A. G. J. Am. Chem. Soc. 2017, 139, 5688-5691. [DOI: 10.1021/jacs.7b03448] Link PDF

38. “Nickel-photoredox catalyzed enantioselective desymmetrization of meso cyclic anhydrides.” Angew. Chem. Int. Ed. 2017, 56, 3679-3683. [DOI:10.1002/anie.201700097] Link PDF

37. “Parameterization of phosphine ligands demonstrates enhancement of nickel catalysis via remote steric effects.” Wu, K.; Doyle, A. G. Nature Chem. 2017, 9, 779-784. [DOI:10.1038/nchem.2741] Link PDF

2016
36. “Direct C(sp3)−H Cross Coupling Enabled by Catalytic Generation of Chlorine Radicals.” Shields, B. J.; Doyle, A. G. J. Am. Chem. Soc. 2016, 138, 12719−12722. [DOI: 10.1021/jacs.6b08397] Link PDF

35. “Nucleophilic (Radio)Fluorination of α-Diazocarbonyl Compounds Enabled by Copper-Catalyzed H–F Insertion.” Gray, E. E.; Nielsen, M. K.; Choquette, K. A.; Kalow, J. A.; Graham, T. J. A.; Doyle, A. G. J. Am. Chem. Soc. 2016, 138, 10802−10805. [DOI: 10.1021/jacs.6b06770] Link PDF

34. “C–H functionalization of amines with aryl halides by nickel-photoredox catalysis.” Ahneman, D. T.; Doyle, A. G. Chem. Sci. 2016, 7, 7002-7006. [DOI:10.1039/C6SC02815B] Link PDF

33. “Nickel-catalyzed enantioselective arylation of pyridine.” Lutz, J. P.; Chau, S. T.; Doyle, A. G. Chem. Sci. 2016, 7, 4105-4109. [DOI: 10.1039/C6SC00702C] Link PDF

32. “Direct Acylation of C(sp3)−H Bonds Enabled by Nickel and Photoredox Catalysis.” Joe, C. L.; Doyle, A. G. Angew. Chem. Int. Ed. 2016, 55, 4040-4043. [DOI: 10.1002/anie.201511438] Link PDF

2015
31. “PyFluor: A Low-Cost, Stable, and Selective Deoxyfluorination Reagent.” Nielsen, M. K.; Ugaz, C. R.; Li, W.; Doyle, A. G. J. Am. Chem. Soc. 2015, 137, 9571−9574. [DOI: 10.1021/jacs.5b06307] Link PDF

30. “Dialkyl Ether Formation via Nickel-Catalyzed Cross Coupling of Acetals and Aryl Iodides.” Arendt, K. M.; Doyle, A. G. Angew. Chem. Int. Ed. 2015, 54, 9876-9880. [DOI: 10.1002/anie.201503936] Link PDF

29. “Electron-Deficient Olefin Ligands Enable Generation of Quaternary Carbons by Ni-Catalyzed Cross Coupling.” Huang, C.-Y.; Doyle, A. G. J. Am. Chem. Soc. 2015, 137, 5638−5641. [DOI: 10.1021/jacs.5b02503] Link PDF

28. “A Modular, Air-Stable Nickel Precatalyst.” Shields, J. D.; Gray, E. E.; Doyle, A. G. Org. Lett. 2015, 17, 2166−2169. [DOI: 10.1021/acs.orglett.5b00766] Link PDF

2014
27. “Merging photoredox with nickel catalysis: Coupling of α-carboxyl sp3-carbons with aryl halides.” Zuo, Z.; Ahneman, D.; Chu, L.; Terrett, J.; Doyle, A. G.; MacMillan, D. W. C. Science 2014, 345, 437-440. [DOI: 10.1126/science.1255525] Link PDF

26. “The Chemistry of Transition Metals with Three-Membered Ring Heterocycles.” Huang, C.-Y.; Doyle, A. G. Chem. Rev. 2014, 114, 8153-8198. [DOI: 10.1021/cr500036t] Link PDF

25. “Enantioselective radiosynthesis of positron emission tomography (PET) tracers containing [18F]fluorohydrins.” Graham, T. J. A.; Lambert, R. F.; Ploessl, K.; Kung, H. F.; Doyle, A. G. J. Am. Chem. Soc. 2014, 136, 5291-5294. [DOI: 10.1021/ja5025645] Link PDF

24. “Mechanistic Investigations of Palladium-Catalyzed Allylic Fluorination.” Katcher, M. H.; Norrby, P.-O.; Doyle, A. G. Organometallics. 2014, 33, 2121-2133. [DOI: 10.1021/om401240p] Link PDF

23. “Enantioselective, Nickel-Catalyzed Suzuki Cross-Coupling of Quinolinium Ions.” Shields, J. D.; Ahneman, D. T.; Graham, T. J. A.; Doyle, A. G. Org. Lett. 2014, 16, 142-145. [DOI: 10.1021/ol4031364] Link PDF

2013
22. “Directed Nickel-Catalyzed Negishi Cross Coupling of Alkyl Aziridines.” Nielsen, D. K.; Huang, C.-Y.; Doyle, A. G. J. Am. Chem. Soc. 2013, 135, 13605–13609. [DOI: 10.1021/ja407223g] Link PDF

21. “Palladium-Catalyzed Allylic C–H Fluorination.” Braun, M.-G.; Doyle, A. G. J. Am. Chem. Soc. 2013, 135, 12990–12993. [DOI: 10.1021/ja407223g] Link PDF

20. “Nickel-Catalyzed Enantioselective Arylation of Pyridinium Ions: Harnessing an Iminium Ion Activation Mode.” Chau, S. T.; Lutz, J. P.; Wu, K.; Doyle, A. G. Angew. Chem. Int. Ed. 2013, 52, 9153-9156. [DOI: 10.1002/anie.201303994] Link PDF

19. “Enantioselective fluoride ring opening of aziridines enabled by cooperative Lewis acid catalysis.” Kalow, J. A.; Doyle, A. G. Tetrahedron 2013, 69, 5702-5709. [DOI: 10.1016/j.tet.2013.01.062] Link PDF

18. “Carbofluorination via a palladium-catalyzed cascade reaction.” Braun, M.-G.;‡ Katcher, M. H.;‡ Doyle, A. G. Chem. Sci. 2013, 4, 1216–1220. (‡ Equal contribution) [DOI: 10.1039/C2SC22198E] Link PDF

2012
17. “Mechanistic Investigation of the Nickel-Catalyzed Suzuki Reaction of N,O-Acetals: Evidence for Boronic Acid-Assisted Oxidative Addition and an Iminium Activation Pathway.” Sylvester, K. T.;‡ Wu, K.;‡ Doyle, A. G. J. Am. Chem. Soc. 2012, 134, 16967-16970. (‡ Equal contribution) [DOI: 10.1021/ja3079362] Link PDF

16. “Synthesis of β-Fluoroamines by Lewis Base Catalyzed Hydrofluorination of Aziridines.” Kalow, J. A.; Schmitt, D. E.; Doyle, A. G. Org. Chem. 2012, 77, 4177–4183. [DOI: 10.1021/jo300433a] Link PDF

15. “Nickel-Catalyzed Negishi Alkylations of Styrenyl Aziridines.” Huang, C.-Y.; Doyle, A. G. J. Am. Chem. Soc. 2012, 134, 9541–9544. [DOI: 10.1021/ja3013825] Link PDF

14. “Nickel-Catalyzed Cross-Coupling of Chromene Acetals and Boronic Acids.” Graham, T. J. A.; Doyle, A. G. Org. Lett. 2012, 14, 1616–1619. [DOI: 10.1021/ol300364s] Link PDF

13. “Fluoride Ring-Opening Kinetic Resolution of Terminal Epoxides: Preparation of (S)-2-Fluoro-1-Phenylethanol.” Shaw, T. W.; Kalow, J. A.; Doyle, A. G. Org. Synth. 2012, 89, 9–18. [DOI: 10.15227/orgsyn.089.0009] Link PDF

2011
12. “Palladium-Catalyzed Regio- and Enantioselective Fluorination of Acyclic Allylic Halides.” Katcher, M. H.; Sha, A.; Doyle, A. G. J. Am. Chem. Soc. 2011, 133, 15902–15905. [DOI: 10.1021/ja206960k] Link PDF

11. “Mechanistic Investigations of Cooperative Catalysis in the Enantioselective Fluorination of Epoxides.” Kalow, J. A.; Doyle, A. G. J. Am. Chem. Soc. 2011, 133, 16001–16012. [DOI: 10.1021/ja207256s] Link PDF

10. “Nickel-Catalyzed Cross Coupling of Styrenyl Epoxides with Boronic Acids.” Nielsen, D. K.; Doyle, A. G. Angew. Chem., Int. Ed. 2011, 50, 6056–6059. [DOI: 10.1002/anie.201101191] Link PDF

9. “Transition Metal-Catalyzed Cross Coupling with N-Acyliminium Ions Derived from Quinolines and Isoquinolines.” Graham, T. J. A.; Shields, J. D.; Doyle, A. G.Chem. Sci. 2011, 2, 980–984. [DOI: 10.1039/c1sc00026h] Link PDF

2010
8. “Palladium-Catalyzed Asymmetric Synthesis of Allylic Fluorides.” Katcher, M. H.; Doyle, A. G. J. Am. Chem. Soc. 2010, 132, 17402–17404. [DOI: 10.1021/jacs.0c03125] Link PDF

7. “Enantioselective Ring Opening of Epoxides by Fluoride Anion Promoted by a Cooperative Dual Catalyst System.” Kalow, J. A.; Doyle, A. G. J. Am. Chem. Soc. 2010, 132, 3268–3269. [DOI: 10.1021/ja100161d] Link PDF

Prior Publications
6. “Enantioselective Thiourea-Catalyzed Additions to Oxocarbenium Ions.” Reisman, S. E.; Doyle, A. G.; Jacobsen, E. N. J. Am. Chem. Soc. 2008, 130, 7198–7199. [DOI: 10.1021/ja801514m] Link PDF
5. “Small-Molecule H-Bond Donors in Asymmetric Catalysis.” Doyle, A. G.; Jacobsen, E. N. Chem. Rev. 2007, 107, 5713–5743. [DOI: 10.1021/cr068373r] Link PDF
4. “Enantioselective Alkylation of Acyclic α,α-Disubstituted Tributyltin Enolates Catalyzed by a Cr(salen) Complex.” Doyle, A. G.; Jacobsen, E. N. Angew. Chem. Int. Ed. 2007, 46, 3701–3705. [DOI: 10.1002/anie.200604901] Link PDF
3. “Enantioselective Alkylations of Tributyltin Enolates Catalyzed by Cr(salen)Cl: Access to Enantiomerically Enriched All-Carbon Quaternary Centers.” Doyle, A. G.; Jacobsen, E. N. J. Am. Chem. Soc. 2005, 127, 62–63. [DOI: 10.1021/ja043601p] Link PDF
2.“C-Arylglucoside synthesis: triisopropylsilane as a selective reagent for the reduction of an anomeric C-phenyl ketal.” Ellsworth, B. A.; Doyle, A. G.; Patel, M.; Caceres-Cortes, J.; Meng, W.; Deshpande, P. P.; Pullockaran, A.; Washburn, W. N. Tetrahedron: Asymmetry 2003, 14, 3243–3247 [DOI: 10.1016/j.tetasy.2003.08.007] Link PDF
1.“A Synthetically Useful, Self-Assembling MMO Mimic System for Catalytic Alkene Epoxidation with Aqueous H2O2.” White, M. C.; Doyle, A. G.; Jacobsen, E. N. J. Am. Chem. Soc. 2001, 123, 7194–7195 [DOI: 10.1021/ja015884g] Link PDF


